Author: CustomCannaPacks Compliance Team Last Updated: February 4, 2026 Estimated Reading Time: 15-18 Minutes Category: Regulatory Compliance & Resources
Introduction: Navigating the Regulatory Realities of Cannabis Packaging in 2026
As the United States cannabis market continues to mature, the era of regulatory ambiguity is widely considered over. Regulatory enforcement surrounding packaging and labeling has shifted from loosely defined guidelines to rigid, actively enforced statutory frameworks. In 2026, packaging compliance is no longer a secondary operational concern—it is one of the most common causes of product recalls, significant financial penalties, and license suspensions across both medical and adult-use markets.
For multi-state operators (MSOs), manufacturers, and emerging brands alike, the compliance challenge is compounded by a fundamental regulatory paradox: cannabis remains illegal at the federal level, yet is heavily regulated at the state level. This fragmented structure creates significant legal exposure for businesses that fail to align their packaging with state-specific requirements.
This guide is intended to serve as an authoritative reference on U.S. cannabis packaging regulations as they stand in 2026. It examines the interaction between federal oversight and state authority, technical standards for child-resistant (CR) packaging, mandatory labeling requirements, material safety considerations, and documentation best practices.
Legal Disclaimer: This content is provided for informational and educational purposes only and does not constitute legal advice. Cannabis regulations are dynamic and subject to frequent amendments. Operators should consult qualified legal counsel and state regulatory agencies (such as the DCC, OCM, or CRC) for the most current requirements in their specific jurisdiction.
1. Federal Oversight vs. State Regulatory Authority
1.1 The Federal Paradox
Despite widespread state-level legalization, cannabis remains classified as a Schedule I controlled substance under the U.S. Controlled Substances Act (CSA). As a result, there is no unified “Federal Cannabis Packaging Act.” However, operators must understand that this absence of direct federal regulation does not eliminate federal oversight entirely.
Several federal agencies indirectly, yet powerfully, shape cannabis packaging compliance:
- Consumer Product Safety Commission (CPSC): The CPSC enforces the Poison Prevention Packaging Act (PPPA) of 1970. While the PPPA was originally designed for pharmaceuticals and household chemicals, nearly all legal cannabis states explicitly reference PPPA testing standards—specifically 16 CFR § 1700.20—as the non-negotiable foundation for child-resistant packaging requirements. If your packaging fails this federal standard, it is non-compliant in virtually every US state.
- Federal Trade Commission (FTC): The FTC regulates truth-in-advertising standards. Packaging that contains misleading claims, unsubstantiated health benefits (e.g., “Cures Insomnia” without FDA approval), or deceptive branding is subject to federal enforcement regardless of state cannabis legality. The FTC has actively issued warning letters to CBD and Delta-8 companies for such violations.
- Food and Drug Administration (FDA): While the FDA generally refrains from regulating state-legal THC products, it actively intervenes in cases involving food mimicry (copying popular snack brands), misbranding, or packaging that poses a clear risk of accidental ingestion by children.
1.2 State-Level Regulatory Primacy
In practice, the day-to-day reality of cannabis packaging compliance is dictated almost entirely at the state level. Each legal market is governed by its own regulatory authority—such as California’s Department of Cannabis Control (DCC), New York’s Office of Cannabis Management (OCM), or Michigan’s Cannabis Regulatory Agency (CRA)—with unique statutory codes.
Critically, compliance is not portable. A packaging solution that is fully compliant in Colorado may be deemed “misbranded” or even illegal contraband in Massachusetts due to granular differences in:
- Universal THC Symbol: States mandate different designs, colors, and minimum sizes (e.g., 0.5″ x 0.5″).
- Warning Language: Some states require rotating health warnings; others require specific, verbatim paragraphs about driving under the influence.
- Font Sizes: Minimum font size requirements for the “Net Weight” or “Universal Symbol” vary (e.g., 6 point vs. 1/16th inch).
- Opacity Requirements: States like Hawaii or Utah may require packaging to be fully opaque, while others allow clear windows.
- Resealability Mandates: Rules differ on which product types require resealable child-resistance.
Operators distributing across multiple states must ensure packaging is customized to each jurisdiction’s requirements, often necessitating distinct SKU management for the same product.
2. Child-Resistant Packaging: Technical and Legal Standards
Child-resistant (CR) packaging is the cornerstone of cannabis compliance and the most heavily scrutinized component during inspections and audits.
Why Child-Resistant Packaging Is the Primary Enforcement Trigger
Regulators view non-compliant packaging as an immediate public safety threat to minors. Unlike minor administrative errors, a failure in child-resistance often results in instant operational shutdowns or product seizures rather than simple warnings.
2.1 The Governing Standard: 16 CFR § 1700.20
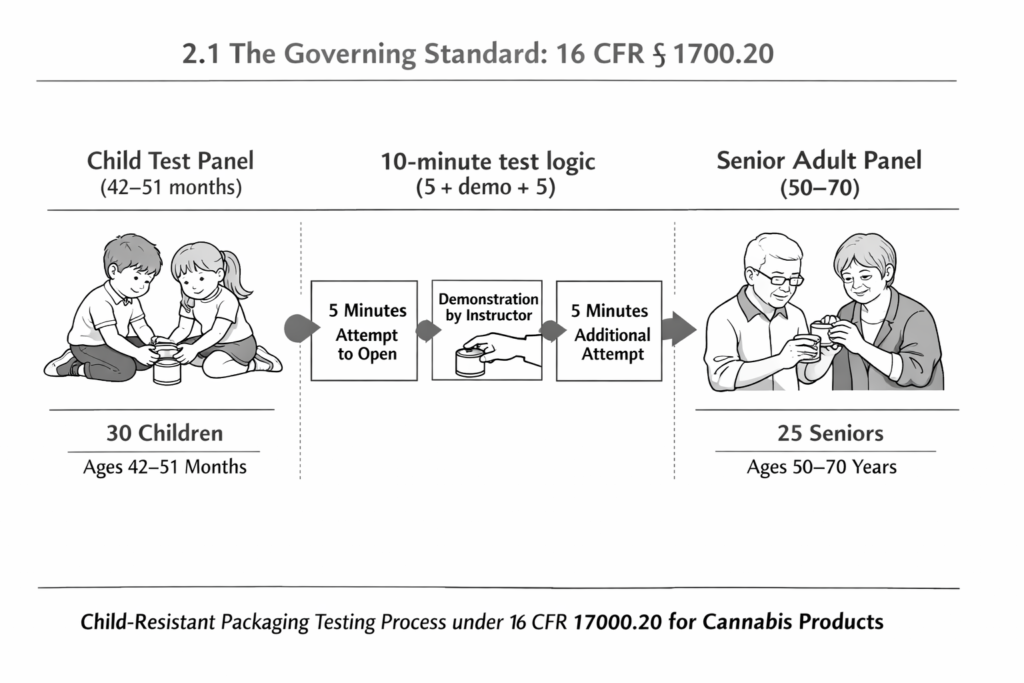
When state regulations mandate “child-resistant packaging,” they are legally referring to compliance with Title 16, Code of Federal Regulations, Section 1700.20, which defines standardized testing protocols.
Certification is not a self-declared status; it requires testing by an accredited third-party laboratory using two specific demographic panels:
- Child Test Panel (42–51 months old):
- Children are given 5 minutes to try and open the package.
- If they fail, they are shown a visual demonstration of how to open it.
- They are given another 5 minutes.
- Requirement: At least 80% of children must be unable to open the package within the total 10 minutes.
- Senior Adult Test Panel (50–70 years old):
- Adults are tested to ensure the package is not too difficult for legitimate consumers to access.
- Requirement: At least 90% of adults must be able to open and properly close the package within a 5-minute period (and within 1 minute for a second attempt).
Packaging that fails either the child protection criterion OR the senior accessibility criterion is deemed non-compliant.
2.2 Lifetime vs. Initial Child Resistance
Regulations typically distinguish between two operational categories of child resistance:
- Lifetime (Resealable) Child Resistance: Required for multi-serving products such as flower (3.5g jars/bags), multi-dose edibles (gummies), or vape cartridges. The packaging must remain child-resistant after repeated openings.
- Technologies: CR Zippers (Press-to-Close), Push-and-Turn Caps, Squeeze-and-Turn Jars.
- Initial (Single-Use) Child Resistance: Permitted for single-serving products intended to be consumed immediately upon opening (e.g., a single infused beverage can or a single brownie). Once opened, the child-resistant feature is rendered void. This is subject to strict state approval.
2.3 Tamper-Evident Features
Child resistance alone is often insufficient. Most states (including CA, CO, WA, OR) also require tamper-evident features that clearly indicate to the consumer whether a package has been opened or manipulated prior to purchase.
Common Tamper-Evident Mechanisms:
- Shrink Bands: Plastic seals around the neck of a jar or bottle.
- Breakaway Caps: Plastic rings that detach upon first opening (similar to soda bottles).
- Induction Seals: Foil liners sealed to the rim of a container.
- Security Labels: “Void” tapes or stickers that tear/leave residue if peeled.
3. Cannabis Packaging Labeling Requirements
Labeling violations represent the most common compliance failure due to the extreme specificity and variability of state rules. A label is not just branding; it is a legal document attached to the product.
Why Labeling Errors Are the Leading Cause of Cannabis Product Recalls
Unlike physical packaging defects, labeling errors—such as incorrect THC potency claims, missing allergens, or omitted warning symbols—are easily auditable and legally require immediate removal from shelves to protect consumers.
3.1 Label Panel Structure
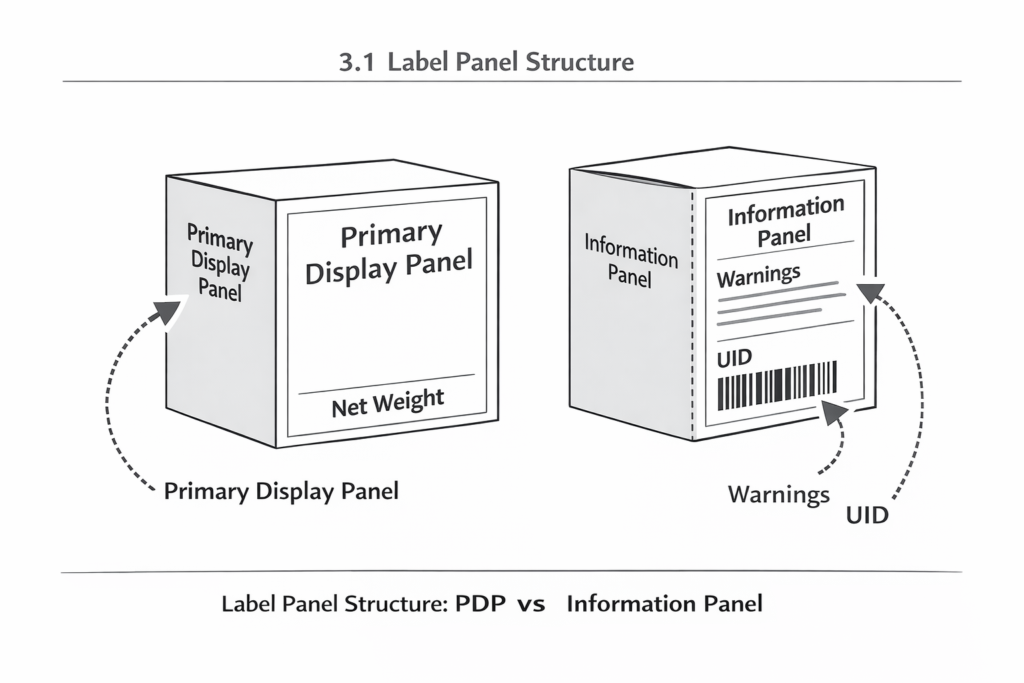
Regulations often distinguish between panels to ensure critical information is prioritized:
- Primary Display Panel (PDP): The part of the label most likely to be displayed to the consumer at retail. It typically must contain:
- Product Identity: Generic name (e.g., “Cannabis Infused Gummy”).
- Net Quantity of Contents: Expressed in both U.S. Customary (oz) and Metric (g/ml) units.
- Universal Symbol: Often required in the corner of the PDP.
- Information Panel: The secondary area (back or side) containing:
- Statutory Warnings.
- Ingredient Lists (descending order by weight).
- Manufacturer/Distributor License Numbers.
- Batch/UID Data.
3.2 Universal THC Symbols

Most states require a government-issued universal cannabis symbol to alert consumers that the product contains THC. These symbols must adhere to precise size, color, and placement rules.
- California: A black triangle with a cannabis leaf and an exclamation point (!), containing the text “CA” and “THC”.
- Michigan: An inverted triangle with a red marijuana leaf and “!”
- Massachusetts: A red triangle containing a black cannabis leaf icon and text “CONTAINS THC”.
Compliance Tip: Failure to meet minimum size specifications (e.g., printing the symbol too small to save design space) is grounds for immediate product rejection or recall.
3.3 Mandatory Warning Statements
States mandate exact warning language, often requiring verbatim reproduction including capitalization and bolding.
Common Warning Themes:
- Age Restriction: “For use only by adults 21 years of age and older.”
- Impairment: “Cannabis creates intoxicating effects and may be habit-forming.”
- Health Risks: “There may be health risks associated with consumption of this product.”
- Pregnancy/Breastfeeding: Warnings against use by pregnant or breastfeeding women.
- Driving: “Do not drive or operate machinery while under the influence of this product.”
3.4 Traceability and Batch Identification
To facilitate recalls and regulatory audits, every single unit of packaging must display traceable identifiers tied to the state’s seed-to-sale tracking system (such as Metrc or BioTrack). Required data points usually include:
- UID / Tag Number.
- Batch or Lot Number.
- Date of Harvest / Manufacturing / Packaging.
- Full Cannabinoid Profile (THC/CBD/Terpenes per serving and per package).
4. Prohibited Packaging Practices
Understanding what is banned is as critical as understanding what is required. Regulators are aggressive in eliminating packaging that appeals to minors.
4.1 Appeal to Children (The “Reasonable Person” Standard)
Packaging is strictly prohibited from bearing any resemblance to commercially available candy, cartoons, or toys. Prohibited elements commonly include:
- Cartoons: Any drawing or caricature of a human, animal, or creature.
- Candy Branding: Use of words like “Candy,” “Taffy,” “Lollipop,” or “Gummy Bear” (states often prefer “Fruit Chew” or “Lozenge”).
- Bright Neon Colors: Some states restrict the color palette to prevent “eye-catching” appeal to toddlers.
- Shape: Edibles cannot be shaped like humans, animals, or fruits.
4.2 Opaque Requirements
Many states (e.g., Colorado, Nevada) require that the actual cannabis product (especially edibles and concentrates) not be visible through the packaging at the point of retail exit. This sometimes necessitates placing a transparent jar inside an opaque box or using an opaque “Exit Bag” at the dispensary counter.
5. Packaging Materials and Product Stability
Compliance extends beyond the label to the physical chemistry of the packaging materials.
5.1 Food-Grade and Safety Standards
Any material in direct contact with cannabis products (especially edibles and flower) must meet FDA food-contact standards under 21 CFR § 177. Common compliant materials include:
- Glass: Inert and impermeable.
- PET (Polyethylene Terephthalate): Durable, clear, and lightweight.
- PP (Polypropylene): High chemical resistance, commonly used for caps and liners.
- Mylar (Multi-layer films): Often utilized for barrier bags.
5.2 Chemical Compatibility & Leaching
Cannabis extracts (particularly terpenes) are chemically aggressive solvents. Inferior plastics can degrade, warp, or leach chemicals into the product.
- Concentrates: Glass jars with PTFE (Teflon) or F217 liners are the industry standard to prevent terpene erosion of the cap liner.
- Vape Carts: Must pass heavy metal testing; packaging must ensure no contamination occurs during storage.
5.3 Environmental Protection
- UV Light: Degrades THC into CBN, reducing potency. Amber glass, opaque plastics, or UV-coated films are recommended.
- Oxygen & Moisture: Flower requires humidity control (typically 58-62% RH). Mylar bags with high barrier properties (low Oxygen Transmission Rate – OTR) or jars with airtight seals are essential for maintaining shelf life and preventing mold growth.
6. Testing, Certification, and Documentation
Successful compliance relies on a robust “Paper Trail.” Brands must maintain rigorous records to survive inspections.
6.1 General Certificate of Conformity (GCC)
Every child-resistant packaging SKU used by a cannabis operator must be accompanied by a General Certificate of Conformity (GCC). This document attests that the specific packaging mechanism has passed the 16 CFR § 1700.20 testing protocols.
- Best Practice: Manufacturers should provide this certificate upon request. Retailers and distributors will often demand a GCC before onboarding a new product line.
6.2 ASTM Standards
While ASTM standards are technically voluntary, they are widely recognized by regulators and testing laboratories as the industry benchmark for packaging quality and durability.
- ASTM D3475: Standard Classification of Child-Resistant Packages.
- ASTM D8197: Standard Specification for Maintaining Child Resistance of Packaging. Adhering to ASTM standards helps demonstrate “Good Faith” efforts in compliance and quality assurance.
Conclusion: Compliance as a Strategic Asset
In the regulated cannabis industry, packaging is not merely a container—it is a legal safeguard and a brand ambassador. The cost of compliant packaging is minimal compared to the catastrophic financial and operational risks of non-compliance.
As enforcement intensifies in 2026 and beyond, and as states continue to harmonize their sustainability and safety rules, businesses that treat packaging compliance as a strategic investment rather than a logistical hurdle will be best positioned for long-term stability and market leadership.
This guide reflects regulatory conditions as of early 2026 and will be updated as state and federal guidance evolves.
About the Author
The CustomCannaPacks Compliance Team specializes in cannabis packaging regulations, child-resistant certification standards, and state-level compliance requirements. Our team works closely with packaging manufacturers and operators across regulated U.S. cannabis markets to ensure safety and legal adherence.

